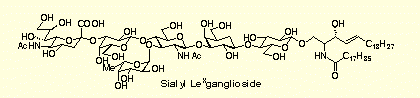Project-glyco
Oligosaccharide Synthesis Based on New Glycosidation
Method
Reviews
- A Stereocontrolled Construction of 2-Azido-2-deoxy-1,2-cis-α-galactosidic Linkages Utilizing 2-Azido-4,6-O-benzylidene-2-deoxygalactopyranosyl Diphenyl Phosphates: Stereoselective Synthesis of Mucin Core 5 and Core 7 Structures.
K. Kakita, T. Tsuda, N. Suzuki, S. Nakamura, H. Nambu, and S. Hashimoto, Tetrahedron., 2012, 68, 5005-5017. [DOI]
- Stereocontrolled Construction of 1,2-cis-α-Glycosidic Linkages Using Glycosyl Diphenyl Phosphates and Synthesis of α-galactosylceramide KRN7000.
H. Nambu, S. Nakamura, N. Suzuki, and S. Hashimoto, Trends Glycosci. Glycotechnol, 2010, 22 (123), 26-40.
- Phosphates, Phosphites and Other O-P Derivatives.
S. Nakamura, H. Nambu, and S. Hashimoto, in Handbook of Chemical Glycosylation: Advances in Stereoselectivity and Therapeutic Relevance (Ed.: A. V. Demchenko), Wiley-VCH, Weinheim, 2008, pp. 223-259.
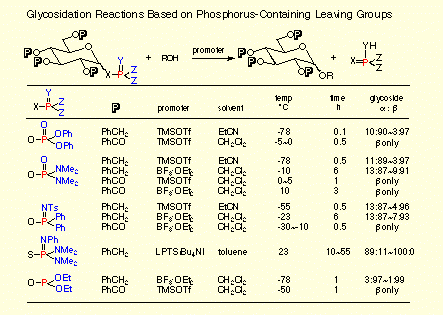
- Glycosidation Reactions Capitalizing on Phosphorus-containing Leaving Groups.
S. Hashimoto and S. Nakamura, in Glycoforum, Glycoword-Glycotechnology
(Ed.: O. Kanie),
http://www.glycoforum.gr.jp/science/word/glycotechnology/GT-E01J.html,
1999.
Selected Publications
- Glycosylation with 2-Acetoamide-2-deoxyglycosyl Donors at a Low Temperature: Scope of the Non-Oxazoline Method
R. Arihara, K. Kakita, N. Suzuki, S. Nakamura, S. Hashimoto, J. Org. Chem., 2015, 88, 4259-4277. [DOI]
- Synthesis of the Tetrasaccharide Repeating Unit from Acinetobacter baumannii Sergroup 018 Capitalizing on Phosphorus-Containing Leaving Groups
R. Arihara, K. Kakita, K. Yamada, S. Nakamura, S. Hashimoto, J. Org. Chem., 2015, 80, 4278-4288. [DOI]
- Direct and Stereoselective Synthesis of 2-Azido-2-deoxy-β-D-Mannosides Using the Phosphate Method.
S. Nakamura, T. Tsuda, N. Suzuki, and S. Hashimoto, Heterocycles, 2009, 77, 843-848. [Abstract]
- Catalytic Stereoselective Glycosidation with Glycosyl Diphenyl Phosphates: Rapid Construction of 1,2-cis-α-Glycosidic Linkages.
M. Koshiba, N. Suzuki, R. Arihara, T. Tsuda, H. Nambu, S. Nakamura, and S. Hashimoto, Chem. Asian J., 2008, 3, 1664-1677. [DOI]
- Direct and Stereoselective Synthesis of β-D-Mannosides Using 4,6-O-Benzylidene-Protected Mannosyl Diethyl Phosphite as a Donor.
T. Tsuda, R. Arihara, S. Sato, M. Koshiba, S. Nakamura, and S. Hashimoto, Tetrahedron, 2005, 61, 10719-10733. [DOI]
- Direct and Stereoselective Synthesis of 2-Acetamido-2-deoxy-β-D-glycopyranosides by Using the Phosphite Method.
R. Arihara, S. Nakamura, and S. Hashimoto, Angew. Chem. Int. Ed., 2005, 44, 2245-2249. [DOI]
- A Highly Stereoselective Construction of 1,2-trans-β-Glycosidic Linkages Capitalizing on 2-Azido-2-deoxy-D-glycosyl Diphenyl Phosphates as Glycosyl Donors.
T. Tsuda, S. Nakamura, and S. Hashimoto, Tetrahedron, 2004, 60, 10711-10737. [DOI]
- A Stereocontrolled Construction of 2-Azido-2-deoxy-1,2-trans-β-glycosidic Linkages Utilizing 2-Azido-2-deoxyglycopyranosyl Diphenyl Phosphates.
T. Tsuda, S. Nakamura, and S. Hashimoto, Tetrahedron Lett., 2003, 44, 6453-6457. [DOI]
- Direct and Stereoselective Construction of β-Mannosidic Linkages Capitalizing on 4,6-O-Benzylidene-Protected D-Mannopyranosyl Diethyl Phosphite.
T. Tsuda, S. Sato, S. Nakamura, and S. Hashimoto, Heterocycles, 2003, 59, 509-515. [Abstract]
- Chemoselective Glycosidation Strategy Based on Glycosyl Donors and Acceptors Carrying Phosphorus-Containing Leaving Groups: A Convergent Synthesis of Ganglioside GM3.
H. Sakamoto, S. Nakamura, T. Tsuda, and S. Hashimoto, Tetrahedron Lett., 2000, 41, 7691-7695. [DOI]
- A Facile Synthesis of 3'-Deoxy-3'-fluorothymidine via a Highly Stereoselective Glycosylation with 2,3-Dideoxy-3-fluoro-D-erythro-pentofuranosyl Diethyl Phosphite.
J. Inagaki, H. Sakamoto, M. Nakajima, S. Nakamura, and S. Hashimoto, Synlett, 1999, 1274-1276. [DOI]
- An Extremely Mild and Stereocontrolled Construction of 1,2-cis-α-Glycosidic Linkages via Benzyl-Protected Glycopyranosyl Diethyl Phosphites.
H. Tanaka, H. Sakamoto, A. Sano, S. Nakamura, M. Nakajima, and S. Hashimoto, Chem. Commun., 1999, 1259-1260. [DOI]
- N-Glycosylation with Glycosyl Diethyl Phosphites: A Highly Stereoselective Synthesis of 2'-Deoxy-β-ribonucleosides.
S. Hashimoto, J. Inagaki, H. Sakamoto, A. Sano, and M. Nakajima, Heterocycles, 1997, 46, 215-220. [Abstract]
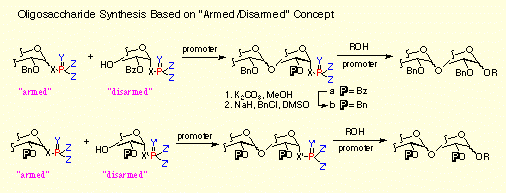
- "Armed-Disarmed" Glycosidation Strategy Based on Glycosyl Donors and Acceptors Carrying Phosphoroamidate as a Leaving Group: A Convergent Synthesis of Globotriaosylceramide.
S. Hashimoto, H. Sakamoto, T. Honda, H. Abe, S. Nakamura and S. Ikegami, Tetrahedron Lett., 1997, 38, 8969-8972. [DOI]
- Oligosaccharide Synthesis Based on Glycosyl Donors and Acceptors Carrying Phosphorus-Containing Leaving Groups.
S. Hashimoto, H. Sakamoto, T. Honda, and S. Ikegami, Tetrahedron Lett., 1997, 38, 5181-5184. [DOI]
- An Attempt at the Direct Construction of 2-Deoxy-β-glycosidic Linkages Capitalizing on 2-Deoxyglycopyranosyl Diethyl Phosphites as Glycosyl Donors.
S. Hashimoto, A. Sano, H. Sakamoto, M. Nakajima, Y. Yanagiya, S. Ikegami, Synlett, 1995, 1271-1273. [DOI]
- An Extremely Mild and General Method for the Construction of 1,2-trans-β-Glycosidic Linkages via Glycopyranosyl Diethyl Phosphites with Participating Groups at C-2.
S. Hashimoto, A. Sano, K. Umeo, M. Nakajima, S. Ikegami, Chem. Pharm. Bull., 1995, 43, 2267-2269. [Journal@rchive]
- An Extremely Mild and Stereocontrolled Construction of 1,2-trans-β-Glycosidic Linkages Capitalizing on Benzyl-Protected Glycopyranosyl Diethyl Phosphites as Glycosyl Donors.
S. Hashimoto, K. Umeo, A. Sano, N. Watanabe, M. Nakajima, and S. Ikegami, Tetrahedron Lett., 1995, 36, 2251-2254. [DOI]
- A Stereocontrolled Construction of 2-Deoxy-β-glycosidic Linkages via 1,2-trans-β-Glycosidation of 2-Deoxy-2-[(p-methoxyphenyl)thio]glycopyranosyl N,N,N',N'-Tetramethylphosphoroamidates.
S. Hashimoto, Y. Yanagiya, T. Honda, and S. Ikegami, Chem. Lett., 1992, 1511-1514. [Journal@rchive]
- An Efficient Construction of 1,2-trans-β-Glycosidic Linkages Capitalizing on Glycopyranosyl N,N,N',N'-Tetramethylphosphoroamidates as Shelf-Stable Glycosyl Donors.
S. Hashimoto, Y. Yanagiya, T. Honda, H. Harada, and S. Ikegami, Tetrahedron Lett., 1992, 33, 3523-3526. [DOI]
- A New and General Glycosidation Method for Podophyllum Lignan Glycosides.
S. Hashimoto, T. Honda, and S. Ikegami, Tetrahedron Lett., 1991, 32, 1653-1654. [DOI]
- An Extremely Mild and General Method for the Stereocontrolled Construction of 1,2-cis-Glycosidic Linkages via S-Glycopyranosyl Phosphorodiamidimidothioates.
S. Hashimoto, T. Honda, and S. Ikegami, Tetrahedron Lett., 1990, 31, 4769-4772. [DOI]
- An Efficient Construction of 1,2-trans-β-glycosidic Linkages via Benzyl-Protected Glycopyranosyl P,P-Diphenyl-N-(p-Toluenesulfonyl)phosphinimidates.
S. Hashimoto, T. Honda, and S. Ikegami, Chem. Pharm. Bull., 1990, 38, 2323-2325. [Journal@rchive]
- A Mild and Rapid 1,2-trans-Glycosidation Method via Benzoyl-Protected Glycopyranosyl P,P-Diphenyl-N-(p-Toluenesulfonyl)phosphinimidates.
S. Hashimoto, T. Honda, and S. Ikegami, Heterocycles, 1990, 30, 775-778. [Abstract]
- A Rapid and Efficient Synthesis of 1,2-trans-β-Linked Glycosides via Benzyl- or Benzoyl-Protected Glycopyranosyl Phosphates.
S. Hashimoto, T. Honda, and S. Ikegami, J. Chem. Soc., Chem. Commun., 1989, 685-687. [DOI]
Laboratory of Synthetic and Industrial Chemistry
Faculty of Pharmaceutical Sciences
Hokkaido University
Top Page of Our Laboratory